Partnership Opportunities
Partnership Opportunities at the 5th Gene Therapy Immunogenicity Summit
With a wealth of recent approvals in the gene therapy space and 850+ active trials, the field is moving fast. However, the complexities in predicting, measuring, and mitigating immune responses in both, animal models and patients, threaten developers with serious safety concerns with these bottlenecks leading to clinical holds.
Decision makers from leading drug developers have expressed that there is little visibility into which contract service providers have the experience and expertise to meet these demands. After extensive research with biopharma leaders, there is a clear lack of belief in the quality of two main services:
- Assay Development Tools – effective, validated, and quick assay development tools to effectively measure and monitor immunogenicity in a clinical and preclinical capacity
- Animal Models – appropriate animal models to test and understand immune-related safety and efficacy profiles and improve the translatability of gene therapies when approaching IND-filing and first-in-human trials
Partnering with the 5th Annual Gene Therapy Immunogenicity Summit will bring you face-to-face with 100+ immunology, bioanalytical, toxicology, preclinical, clinical, translational, and scientist leaders in the field, actively seeking networking and collaborating opportunities to overcome the challenges they face.
Your brand, your message, and your reputation – showcased in front of the leading experts in the gene therapy field.
2024 Partner:

Experts Need Your Help With:
- Assay Development Tools – effective, validated and quick assay development tools to effectively measure and monitor immunogenicity in a clinical and preclinical capacity.
- Animal Models – appropriate animal models to test and understand immune-related safety and efficacy profiles and improve the translatability of gene therapies when approaching IND-filing and first-in-human trials
Why Partner?
Meet your 2024 commercial objectives and educate decision-makers on how your expertise can overcome immunogenicity bottlenecks in gene therapy development
Showcase your expertise to leading gene therapy organisations by securing an exhibition booth to set yourself up as the go-to service provider in the gene therapy community
Demonstrate thought leadership and drive brand exposure by securing a slot on the agenda to connect with the community and generate partnerships
Maximize the balance of content and networking to generate leads and build new relationships with industry pioneers
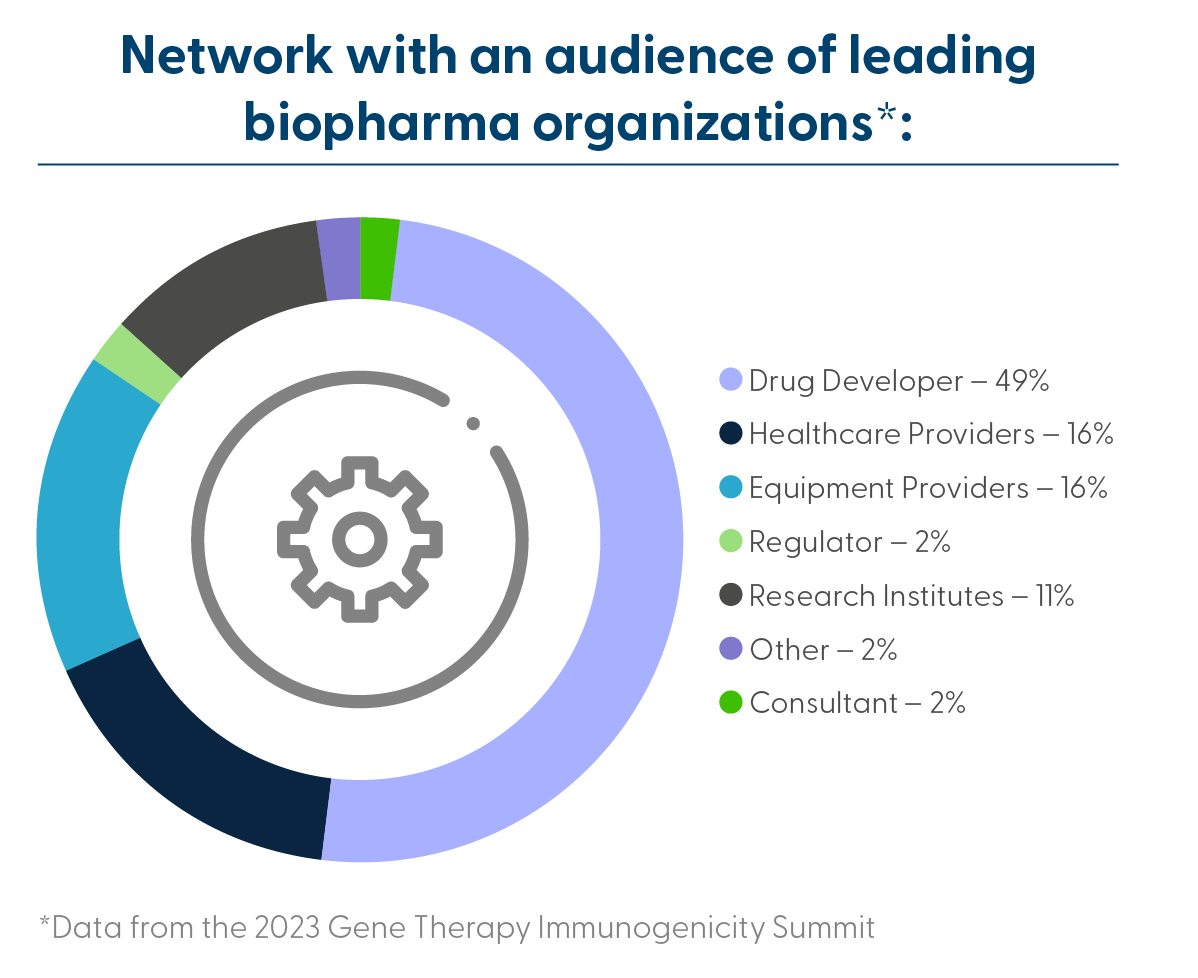
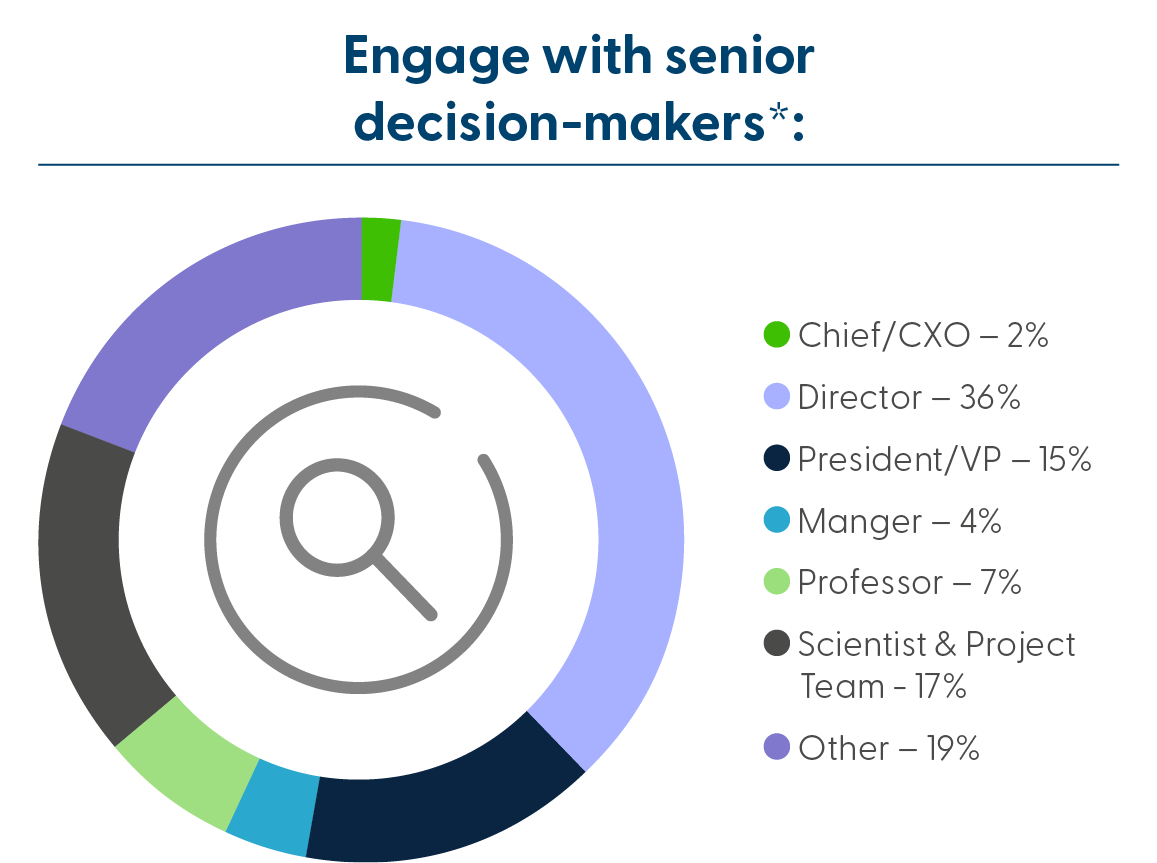
Who Will You Meet:
Past Partners:



